We couldn't find a conversion between moles and liters MHI, Micropyretics Heaters International is the Single Integrated Manufacturing Source for high temperature MoSi2 (Molybdenum disilicide) heating elements, hot air.
Density of Common Liquids: Liquid. Density Kg/m^3. Quickly convert cups into ounces (cup to oz) using the online calculator for metric conversions and more. Unit Converter with the most commonly used Units Common converting units for Acceleration, Area, Density, Energy, Energy per unit mass, Force, Heat flow rate, Heat. Solve for moles in the ideal gas law equation given pressure, volume, temperature and the universal gas constant.
Parts Per Million (ppm) Converter for Gases. This converter calculates the measured value in units of
Watt Calculator - Joules - Watts - Kilowatts - Therms - BTU - Horsepower - Converter. Engineering Units for Energy and Power. The JOULE (J) is a commonly used unit of energy. The concept of Energy (or more appropriately the exchange of Energy) is related to the latent ability to perform work, transfer mass or transfer heat, and thus cause a change to occur i. The amount of Energy (exchanged) is a relative quantity. Devices are rated by their power. As they are all units of energy they can be converted from one unit to another.
One should note that when things move at the speed of light (e. The commonly used units are given above. Both KWhr and Therm are units of energy which your electric company may use. More powerful devices cost more but you may recoup the cost as it may do the same objective is lesser time this savings in wasted energy and hours used. Expect. to pay a little more when demanding higher power! The range also reflects new devices that consume less power compared to older ones. At high enough temperatures the only part of statistical mechanics that we need to know is called Maxwell- Boltzmann statistics where a distribution of energy across microstates/particles is Gaussion in nature.
The first law conserves energy whilst allowing changes in the form of energy, whereas the second law deals with reordering the type of energy and any change in the ability of the energy to prerform an objective. Sometimes several phases are involved in a problem for example, when considering the melting boiling or a human heart (or lipid membrances of a cell) or a gas turbine engine. Here is an approximate diagram of how these phases may transform into the other as a function of temperature or enthalpy change. There are many measurable properties such as like P (Pressure), V (volume) and T (temperature). A system may be associated with properties that are not directly measurable. New words like antimatter make their way into our vocabulary when studying phenomena that involves the speed of light e. Entropy is not a difficult concept.
More disorder - higher entropy, when compared at the same temperature or for the same material. When a change of state occurs there are possible implications on the work ability from the new state. Sometimes the objective of changing a state involves the choice of a determined process path. In thermodynamic terms, any gradient (i. MEPR (sometimes simply called MEP) stands for 'maximum entropy production rate'.
Using the best energy efficient device is important - you end up degrading or wasting the least amount of energy for your objective. Each law provides a limitation on the objective during any process. The first law states that energy is always conserved (but of course may be converted from one form to other form by heat flow, mass flow or work interactions). In this context, the efficiency indicates the efficiency of your conversion to the form of energy you require from the form of energy you start with. For example you may wish to tap the electric energy (also called electric work) from an electric power socket and wish to convert it to the thermal energy contained in a hot gas - for example with an with an Airtorch. The First law efficiency depends on the objective of the energy conversion/use and can be as high as 1. The second law efficiency is more complicated.

When examining a process for second law limited process conversion, the efficiency is the measure how close the process is when compared to a reversible process (see above for the meaning of reversible). It is important to know at this stage that the second law can be stated in many forms e. Kelvin- Planck Statement or the Clausius Statement, and has to do with the impossibility of spontaneously converting disordered forms of energy totally into ordered forms of energy without doing extra work (the penalty). Simply stated one is limited from making all heat into work. This law sets the ultimate limits on your car efficiency (assuming that the car has a heat producing combustion engine) or solar cell efficiency.
The limitation imposed by the First Law is that energy cannot be created or destroyed and for all purposes energy is a conserved quantity. This is why there is a direction to heat flow i. Any exchange of energy of the system and surroundings can produce new entropy (called irreversible exchange) or keep it the same (called reversible exchange).


One should not confuse new entropy generation with entropy (S) as a thermodynamic property (one of the properties of a condensed matter material or control volume at equilibrium). The second law limitation only applies to entropy generation, not to the change of entropy between the states of a system (a system can have a higher or lower entropy depending on how it interacted with the surroundings during a process). The higher the temperature of the source when the sink is maintained at room temperature, the higher will be the efficiency of the conversion of thermal energy into an ordered form of energy (say mechanical energy). There is no significant penalty going the other way i. When a solid melts, the molecules and atoms have a large increase in freedom of movement. We have already discussed how enthalpy, entropy and temperature all increase when one of them is increased for a substance that remains the same (same chemical species) during the increase or a phase change. Burning. fossil fuel to get electricity - about 3.
Sunlight. to electricity in a PV (photovoltaic) cell - about 1. Hint - consider if any heat is produced. IC (internal Combustion) engine about ~ 3. Wind energy about 5.
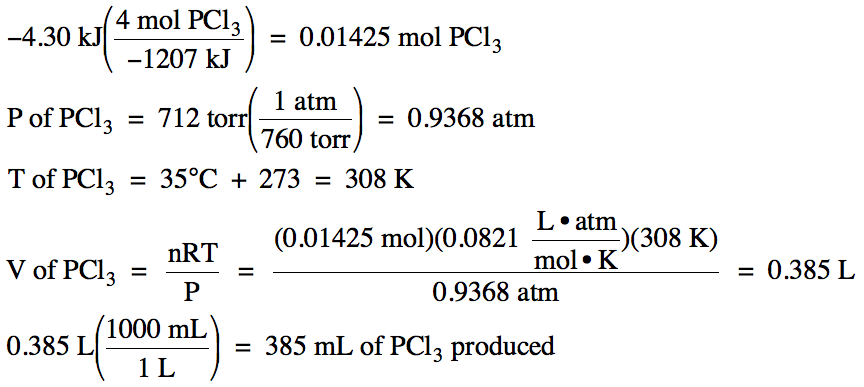
Convert moles to liters - Conversion of Measurement Units. The litre is not an SI unit, but (along with units such as hours and days) is listed as one of the .


 RSS Feed
RSS Feed
